The pharmaceutical industry is a cornerstone of global health, yet the journey of a new drug from concept to market is fraught with immense financial challenges. Understanding the intricacies of r&d costs for new drug development is crucial for pharmaceutical companies, investors, policymakers, and ultimately, patients worldwide. These costs significantly impact drug pricing, accessibility, and innovation rates.
The escalating price of drug development, often cited as exceeding $2.6 billion per approved drug, necessitates a thorough examination of the contributing factors. These include the high failure rate of drug candidates, the extensive regulatory hurdles, the complexity of modern drug targets, and the increasing demand for personalized medicine. Effectively managing these r&d costs for new drug development is paramount for sustaining pharmaceutical innovation.
Successfully navigating this landscape requires a strategic approach to research, development, and commercialization. Accurate cost estimation, efficient clinical trial design, and effective intellectual property management are all vital components. Furthermore, collaboration between industry, academia, and government agencies is essential for sharing resources and mitigating risks associated with r&d costs for new drug development.
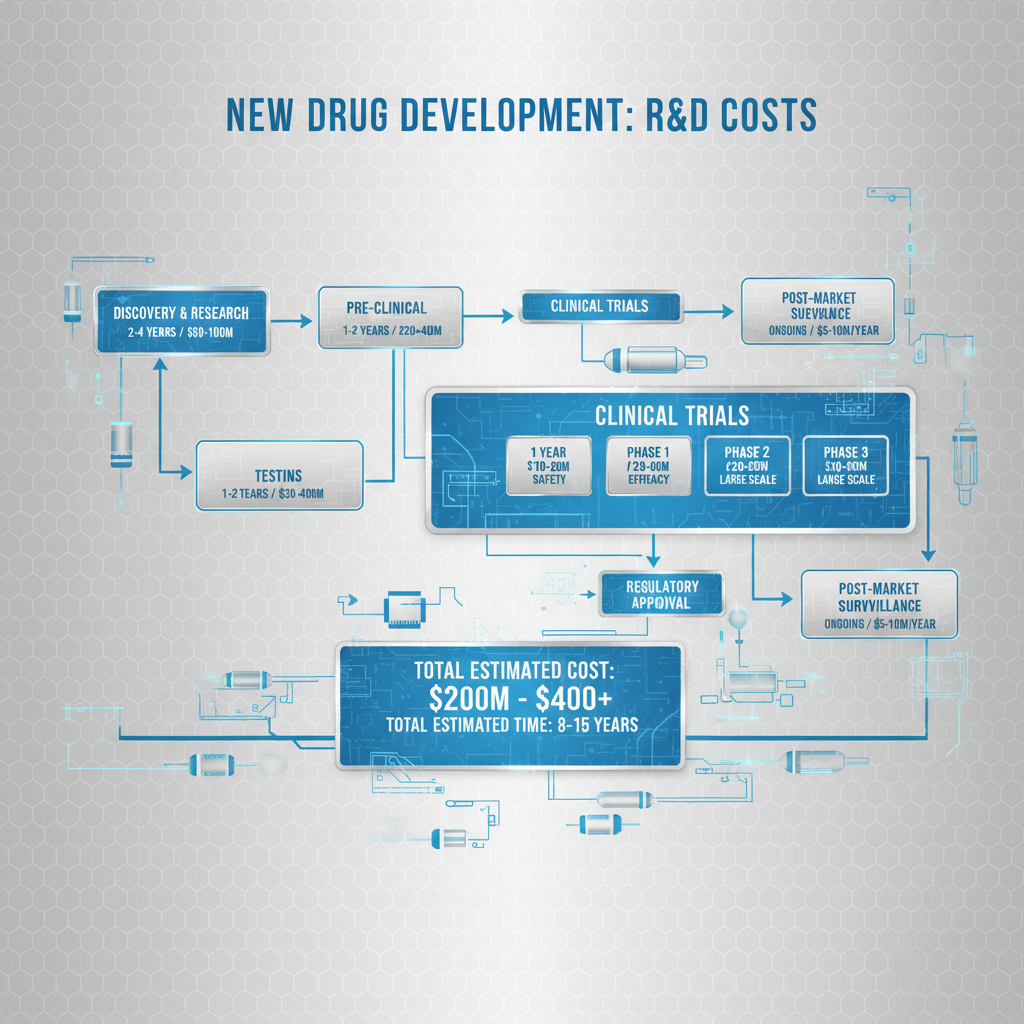

The development of new pharmaceuticals is a long, complex, and extraordinarily expensive undertaking. Understanding the nuances of r&d costs for new drug development is essential for stakeholders across the healthcare ecosystem. These costs encompass everything from initial discovery research to post-market surveillance, and influence the availability and affordability of life-saving medications.
Factors driving these high costs include the increasing stringency of regulatory requirements, the need for larger and more complex clinical trials, and the growing focus on developing treatments for chronic and rare diseases. Efficiently managing these expenses is vital for ensuring a sustainable pipeline of innovative drugs.
r&d costs for new drug development represent the total expenses incurred by a pharmaceutical company or research institution in the process of discovering, developing, and preparing a new drug for market. This includes preclinical research, clinical trials (Phase I, II, and III), regulatory submissions, and post-market surveillance. These costs do not include the expenses associated with marketing and sales after approval.
The process typically begins with basic research to identify potential drug targets and promising compounds. Preclinical studies then evaluate the safety and efficacy of these compounds in laboratory settings and animal models. Successful candidates progress to clinical trials, which involve testing the drug in human volunteers. The entire process can take 10-15 years and involves a significant risk of failure at any stage.
These costs are not merely financial expenditures; they represent an investment in human health and future medical advancements. The development of novel therapies relies on substantial, sustained financial backing to address unmet medical needs.
Several key components contribute significantly to the overall r&d costs for new drug development. Firstly, preclinical research, including target identification, drug discovery, and animal testing, can account for a substantial portion of early-stage expenses. Secondly, clinical trials, especially Phase III trials involving large patient populations, are incredibly expensive to conduct and manage.
Thirdly, regulatory costs, including preparing and submitting documentation to agencies like the FDA or EMA, are considerable. Fourthly, the cost of failed drug candidates – those that do not demonstrate safety or efficacy – represents a significant ‘sunk cost’. Finally, manufacturing scale-up and process development, preparing for commercial production, add substantially to the financial burden.
Effective cost management requires a detailed understanding of these components. Strategic outsourcing, utilizing advanced technologies like artificial intelligence and machine learning for drug discovery, and implementing efficient clinical trial designs can help to mitigate expenses. Prioritizing projects with the highest probability of success and focusing on areas with significant unmet medical needs are also crucial strategies for optimizing r&d costs for new drug development.
Intellectual property protection plays a vital role. Securing patents and maintaining exclusivity are essential for recouping investment and incentivizing further innovation. Conversely, generic drug development, while important for affordability, presents a challenge to innovator companies seeking to maximize returns on their r&d costs for new drug development.
The management of r&d costs for new drug development is a global concern, with varying approaches adopted by different regions and organizations. In the United States, the high cost of healthcare and the stringent regulatory environment drive companies to focus on high-value, innovative therapies. In Europe, collaborative research initiatives and government funding are more prevalent.
Emerging markets, such as China and India, are increasingly becoming important centers for pharmaceutical research and development, often leveraging lower labor costs and a growing pool of skilled scientists. However, these regions also face challenges related to intellectual property protection and regulatory harmonization. International collaborations and partnerships are becoming increasingly common as companies seek to share risks and access diverse expertise.
Organizations like the World Health Organization (WHO) advocate for strategies to reduce r&d costs for new drug development for neglected tropical diseases and essential medicines, particularly in low- and middle-income countries. This often involves public-private partnerships and innovative financing mechanisms.
Optimizing r&d costs for new drug development yields significant benefits. Reduced costs can translate into lower drug prices, improving patient access to essential medicines. Increased efficiency allows companies to invest in a broader range of research projects, potentially leading to the discovery of innovative therapies for previously untreatable conditions.
Furthermore, efficient resource allocation enhances a company’s financial stability and attractiveness to investors. A strong focus on cost management can also foster a culture of innovation and accountability within the organization. Ultimately, optimizing r&d costs for new drug development benefits all stakeholders – patients, companies, and society as a whole.
Several emerging trends are poised to reshape the landscape of r&d costs for new drug development. Artificial intelligence (AI) and machine learning (ML) are increasingly being used to accelerate drug discovery, identify potential drug candidates, and optimize clinical trial design. These technologies can significantly reduce the time and cost associated with these processes.
Another key trend is the growing emphasis on personalized medicine, tailoring treatments to individual patients based on their genetic makeup and other factors. While promising, personalized medicine also presents challenges related to cost and scalability. The adoption of real-world evidence (RWE) and digital health technologies is also expected to play an increasingly important role in drug development and post-market surveillance.
Despite advancements in technology and cost management strategies, significant challenges remain in controlling r&d costs for new drug development. The high failure rate of drug candidates continues to be a major driver of expenses. Increasing regulatory scrutiny and the complexity of modern drug targets also add to the burden.
Innovative solutions include the development of more predictive preclinical models, the use of biomarkers to identify patients most likely to respond to treatment, and the implementation of adaptive clinical trial designs that allow for adjustments based on interim results. Collaboration between industry, academia, and government agencies is also crucial for sharing resources and mitigating risks.
Furthermore, exploring alternative funding models, such as venture philanthropy and public-private partnerships, can help to address the financial challenges associated with developing treatments for neglected diseases and rare conditions.
| Challenge | Impact on r&d Costs | Potential Solution | Expected Cost Reduction |
|---|---|---|---|
| High Drug Failure Rate | Significant sunk costs for failed candidates | Improved target validation & predictive models | 10-15% |
| Complex Clinical Trials | Large patient populations & long durations | Adaptive trial designs & biomarker identification | 15-20% |
| Regulatory Hurdles | Extensive documentation & approval processes | Early engagement with regulatory agencies | 5-10% |
| Data Management & Analysis | High costs associated with data collection & analysis | Implementation of AI & machine learning tools | 8-12% |
| Manufacturing Scale-Up | Challenges in transitioning from lab to commercial production | Early process development & strategic outsourcing | 7-10% |
| Intellectual Property Protection | Legal fees & enforcement costs | Proactive patent strategy & global enforcement | 3-5% |
Several factors contribute to rising costs, including the increasing complexity of drug targets, stringent regulatory requirements, the need for larger and more complex clinical trials, and the pursuit of treatments for chronic and rare diseases. The 'low-hanging fruit' of easily treatable conditions has largely been addressed, leaving more challenging and expensive research areas.
Companies can employ several strategies, such as utilizing artificial intelligence and machine learning for drug discovery, implementing adaptive clinical trial designs, focusing on biomarker-driven patient selection, and fostering collaborations with academic institutions and other pharmaceutical companies to share resources and risks. Prioritizing projects with the highest probability of success is also crucial.
Government regulations, while essential for ensuring patient safety and efficacy, can significantly increase r&d costs for new drug development. Meeting regulatory requirements for clinical trials, data submission, and manufacturing adds substantial expenses. Streamlining regulatory processes and fostering greater international harmonization could help reduce these costs without compromising safety.
AI and machine learning are revolutionizing drug discovery by accelerating the identification of potential drug candidates, predicting drug efficacy and toxicity, and optimizing clinical trial designs. These technologies can significantly reduce the time and cost associated with the early stages of r&d costs for new drug development, although implementation costs and data quality are important considerations.
Generic drug competition significantly reduces the profitability of innovator companies after patent expiration. While generics increase access to affordable medicines, they erode the market share and revenue of the original drug developer. This pressure incentivizes companies to continuously innovate and develop new, patented drugs to maintain their competitive edge and recoup their r&d costs for new drug development.
Collaboration between industry and academia fosters knowledge sharing, leverages complementary expertise, and reduces the financial burden of research. Academia often possesses specialized knowledge and technologies, while industry provides funding and expertise in drug development and commercialization. These partnerships can accelerate innovation and reduce the overall r&d costs for new drug development process.
In conclusion, the r&d costs for new drug development represent a substantial investment in global health, driving innovation and providing solutions to pressing medical needs. Successfully managing these costs requires a multifaceted approach that encompasses technological advancements, strategic partnerships, efficient clinical trial designs, and streamlined regulatory processes. A thorough understanding of the components contributing to these costs is crucial for all stakeholders involved.
Looking ahead, continued investment in research and development, coupled with a commitment to collaboration and innovation, will be essential for addressing future healthcare challenges and ensuring that life-saving medications remain accessible to those who need them. We encourage further exploration of these complex issues and invite you to visit our website at www.hejiachecialtech.com to learn more about our commitment to advancing pharmaceutical innovation.
If you are interested in our products, you can choose to leave your information here, and we will be in touch with you shortly.
